An advanced, CLIA-certified, CAP-accredited, NY CLEP-licensed multiomic services laboratory providing customizable data integrations and fast, accurate testing at competitive prices.
Research Services
Deadline-driven turnaround times
Industry-leading capacity
Advanced lab automation
Competitive pricing

Fast, accurate testing solutions to drive your research forward
Clinical Enterprise can quickly scale test runs, allowing you to get fast, accurate, and consistent results at any volume to drive your research forward.
Our customizable integration options and real-time data exchange can streamline data delivery, providing immediate transfer of results so you can act on your data.
Engineered for integration, powered for collaboration
Eurofins Clinical Enterprise is uniquely positioned to provide the highest throughput for your testing. Our lab’s automation eliminates the margin for error, accelerates data delivery, and delivers consistent results at competitive prices.
Our advanced engineering and IT capabilities allow for a seamless data exchange, allowing you to immediately receive data once it’s ready for analysis.

Consistent, deadline-driven turnaround times
Get accurate, quality data on time, regardless of your project’s scale or testing fluctuations in the lab.

Reliable partnerships
Receive proactive communication from our technical and support teams throughout your project.

Secure data systems
Ensure rapid, secure data exchange and seamless integration with your systems for rapid data delivery.
Our Services

Sequencing Services
Flexible, high-quality next-generation sequencing to rapidly sequence whole genomes, whole exomes, targeted regions, microbiomes, and more.

Genotyping Services
We offer state-of-the-art genotyping arrays using the most modern Illumina Infinium chips available, and qPCR-based testing across a wide range of applications from small- to mid-size (20–50 target) SNP panels.

Customized Testing Services
Need a partner who can work with your specific needs? We provide customized testing, IT integrations, logistics, solutions, and method development.
Our Instruments
High-throughput and purpose-designed automation on advanced equipment
Liquid Handling

- 9 Hamilton STAR Systems
- 3 Agilent BRAVO Systems
- Thermo Fisher KingFisher 96 Flex Systems
- TECAN System for Array
qPCR Systems
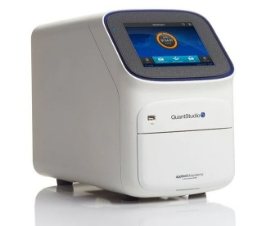
- 6 QuantStudio 5 384-well real-time PCR systems
- QuantStudio 12K with OpenArray™ Technology
Sequencers and Analyzers

- NovaSeq X Plus
- NovaSeq 6000
- 4 Illumina MiSeqs
- 1 Illumina NextSeq 500
- Illumina iSCAN for microarray
- Applied Biosystems™ 3730xl DNA Analyzer for Sanger Sequencing
Why Clinical Enterprise?
- Deadline-driven turnaround times with unparalleled accuracy
- Industry-leading capacity and comprehensive analytical platforms
- CLIA, CAP, and NY CLEP accredited with clinical-grade laboratory-developed services
- Numerous research use only options
- Robust data integration options including HL7, SFTP, API, AWS S3
- LIS-driven real-time reporting at each phase
Ready to move your work forward?
Contact us
Meet with us
Work with us

5 Signs Of A Reputable Reference Laboratory Partner
Download our guide to discover the five signs every researcher should be looking for in a reputable reference laboratory partner.

